-
 Home
Home
-
 News
News
Latest Educational News Stories
Daily update of all national, international news, picture stories, college / university announcements and educational events.
-
 Colleges
Colleges
Pakistan's Largest Database of Colleges and Universities
Explore Largest Directory of Private and Govt. Colleges, Universities and find best institute for your future Education.
-
 Courses
Courses
-
 Admission
Admission
-
 Lectures
Lectures
-
 Online Test
Online Test
Short Question
- 9th Class Physics Short Questions
- 9th Class Chemistry Short Questions
- 9th Class Math Short Questions
- 9th Class Biology Short Questions
- 9th Class Computer Short Questions
- 9th Class English Short Questions
- 10th Class Physics Short Question
- 10th Class Chemistry Short Question
- 10th Class Math Short Question
- 10th Class Biology Short Question
- 10th Class Computer Short Question
- 10th Class English Short Question
-
 Past Papers
Past Papers
-
 Date Sheets
Date Sheets
-
 Results
Results
Exam Results 2024
Check online Results 2024 Matric Inter BA BSc B.Com MA MSc M.Com CSS PCS MCAT ECAT of all educational boards and universities in Pakistan
-
 Study Abroad
Study Abroad
Study Abroad Programs and Opportunities for Pakistani Students
Explore free study abroad search to find programs, consultants, events to study in USA, UK, Australia, China, Malaysia and many others.
-
 Jobs
Jobs
-
 Tutors
Tutors
-
 More
More
-
 Apps
Apps
MCQ's Test For ECAT Chemistry Chapter 8 Chemical Equilibrium
Try The MCQ's Test For ECAT Chemistry Chapter 8 Chemical Equilibrium
-
Total Questions30
-
Time Allowed30
Question # 1
The rate at which a substance reacts is directly proportional to its active mass and the rate of reaction is directly proportional to the product of the active masses of reacting substances, is called
Question # 2
Which one of the following is a buffer
Question # 3
In a reversible chemical reaction having two reactants in equilibrium, if the concentration of the reactants are doubled then the equilibrium constant will
Question # 4


Question # 5
Two moles of HI was heated in a sealed tube at 440°C till the equilibrium was reached. HI was found to be 22% decomposed. The equilibrium constant for dissociation is
Question # 6
Ifkcof a reaction productis verylarge, it indicates that equilibrium occurs :
Question # 7
The solubility of KCIO3salt in water is decreased by adding
Question # 8
A chemical reaction A---------->B is said to be in equilibrium when :
Question # 9
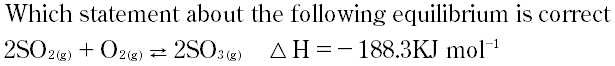
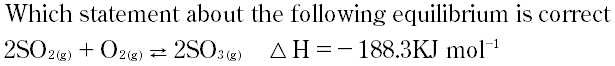
Question # 10
In a reversible reaction, two substances are in equilibrium. If the concentration of each one is reduced to half, the equilibrium constant will be
Question # 11
strength of an acid can be determined by
Question # 12
The substance which increases rate of reaction but remains unchanged at the end of reaction is called :
Question # 13
An excess of aqueous silver nitrate is added to aqueous barium chloride and precipitate is removed by filtration. What are the main ions in the filtrate
Question # 14


Question # 15


Question # 16
H2 + L2 ----2Hl
H2 + L2 ----2HlIn the above equilibrium system, if the concentration of reactants at 25°C is increased, the value KC will :
Question # 17


Question # 18
The rate of a chemical reaction is directly;y proportional to product of molar concentration of reaction substance it is called :
Question # 19
1.1 mol of A is mixed with 2.2 mol of B and the mixture is kept in on litre flask till the equilibrium is reached. At equilibrium, 0.2 mol of C is formed. If the equilibrium reaction is A+2B 2C+D, the value of equilibrium constant is
Question # 20
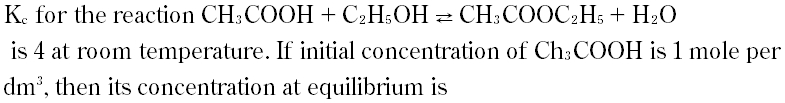

Question # 21
A buffer of a 0.09 molar acetic acid and 0.11 molar sodium acetate has pH = 4.83. If 0.01 mole NaOH in 1 dm3of the buffer solution is added, then pH of the buffer becomes
Question # 22
Law of mass action was given by :
Question # 23


Question # 24
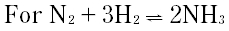
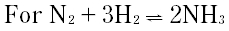
Question # 25
Kspvalue for PbSO4= 1.8 x 10-8mole2dm-6. The maximum concentration of Pb++ions is
Question # 26
The equilibrium constant in a reversible chemical reaction at a given temperature
Question # 27
The solubility of PbF2is 2.6 x 10-3mole dm-3 then its solubility product is
Question # 28
1 mol of N2O4 was decomposed according
to given equation in 1dm3 container. At equilibrium x mole of N2O4
have dissociated. What is the value of KC:
Question # 29
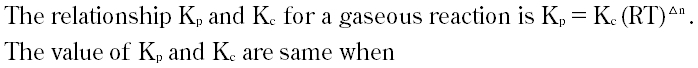
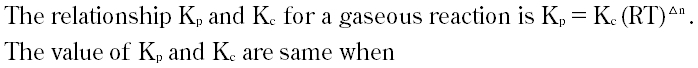
Question # 30
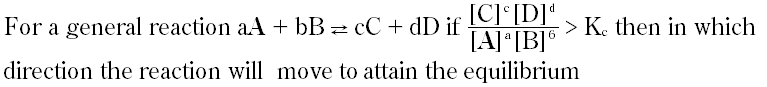
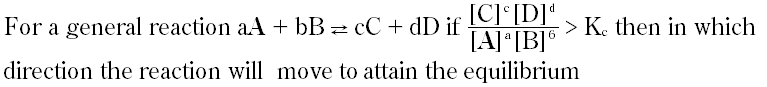
Top Scorers Of ECAT Chemistry Chapter 8 Chemical Equilibrium MCQ`s Test
-
A Arian Ahmed 06 - Mar - 2024 00 Min 04 Sec 115/120 -
H Huzaifa Asim 28 - May - 2024 01 Min 12 Sec 115/120 -
M Muneeb Aslam 03 - Jun - 2024 09 Min 57 Sec 105/120 -
? ?GøÐ øf ?ë??h? 29 - May - 2024 32 Min 31 Sec 95/120 -
H Hamza Malhi 10 - Mar - 2024 14 Min 42 Sec 85/120 -
F Furqan Asif 20 - Mar - 2026 95 Min 59 Sec 84/120 -
M Maryam Fatima 19 - Mar - 2024 09 Min 42 Sec 80/120 -
F Fatima Tabassum 16 - Jan - 2025 09 Min 48 Sec 65/120 -
E Essa raza 22 - Jun - 2024 09 Min 07 Sec 55/120 -
K kayani 21 - Jul - 2024 09 Min 41 Sec 55/120 -
T Tamshi Sohail 20 - Apr - 2024 23 Min 19 Sec 55/120 -
R random 11 - May - 2024 06 Min 15 Sec 50/120 -
H Haniya Ali 14 - Mar - 2024 14 Min 41 Sec 50/120 -
E Ezzah Ansari 15 - May - 2024 15 Min 25 Sec 50/120 -
M Meena 19 - Apr - 2024 10 Min 19 Sec 45/120
ECAT Chemistry Chapter 8 Important MCQ's
| Sr.# | Question | Answer |
|---|---|---|
| 1 |
An excess of aqueous silver nitrate is added to aqueous
barium chloride and precipitate is removed by filtration. What are the main ion
in filtrate? |
A.
Ag+ and NO3- only
B.
Ag+ and Ba2 and NO-3
C.
Ba2 and NO-3
D.
Ba2 and NO-3 and Cl- |
| 2 | For which system does the equilibrium constant, kc has units of (concentration) ? |
A.
N2+3H2-------2NH3
B.
H2+L2---------2HL
C.
2NO2----------N2O4
D.
2HF--------H2+F2 |
| 3 |

|
A. Introduction of an inert gas at constant volume
B. Introduction of PCl3(g) at constant
C. Introduction of PCl5(g) at constant volume
D. Introduction of Cl2at constant volume
|
| 4 | In a reversible reaction, two substances are in equilibrium. If the concentration of each one is reduced to half, the equilibrium constant will be |
A. Reduced to half of its original value
B. Doubled
C. Same
D. Reduced to one fourth its original value
|
| 5 | At certain temperature, 50% of HI is dissociated into H2and I2the equilibrium constant is |
A. 1.0
B. 3.0
C. 0.5
D. 0.25
|
| 6 | An excess of aqueous silver nitrate is added to aqueous barium chloride and precipitate is removed by filtration. What are the main ions in the filtrate | |
| 7 |

|
A. Pressure change
B. Temperature change
C. Concentration change
D. Catalyst
|
| 8 | Base buffer solution can be prepared by mixing |
A. Weak acid and its salt
B. Strong acid and its salt with weak base
C. Weak base and its salt with strong acid
D. Strong base and its salt with weak acid
|
| 9 | A large value of Kcmeans that at equilibrium : |
A. Less reactant and more products.
B. Reactants and product in same amounts.
C. More reactants and less products.
D. None of above.
|
| 10 | A solution having pH = 4 its OH-ion concentration in mole dm-3is |
A. 1.0 x 10-4
B. 1.0 x 10-10
C. 1.0 x 10-14
D. 1 x 100
|








.gif)








Share your comments & questions here
No comments yet. Be the first to comment!