-
 Home
Home
-
 News
News
Latest Educational News Stories
Daily update of all national, international news, picture stories, college / university announcements and educational events.
-
 Colleges
Colleges
Pakistan's Largest Database of Colleges and Universities
Explore Largest Directory of Private and Govt. Colleges, Universities and find best institute for your future Education.
-
 Courses
Courses
-
 Admission
Admission
-
 Lectures
Lectures
-
 Online Test
Online Test
Short Question
- 9th Class Physics Short Questions
- 9th Class Chemistry Short Questions
- 9th Class Math Short Questions
- 9th Class Biology Short Questions
- 9th Class Computer Short Questions
- 9th Class English Short Questions
- 10th Class Physics Short Question
- 10th Class Chemistry Short Question
- 10th Class Math Short Question
- 10th Class Biology Short Question
- 10th Class Computer Short Question
- 10th Class English Short Question
-
 Past Papers
Past Papers
-
 Date Sheets
Date Sheets
-
 Results
Results
Exam Results 2024
Check online Results 2024 Matric Inter BA BSc B.Com MA MSc M.Com CSS PCS MCAT ECAT of all educational boards and universities in Pakistan
-
 Study Abroad
Study Abroad
Study Abroad Programs and Opportunities for Pakistani Students
Explore free study abroad search to find programs, consultants, events to study in USA, UK, Australia, China, Malaysia and many others.
-
 Jobs
Jobs
-
 Tutors
Tutors
-
 More
More
-
 Apps
Apps
MCQ's Test For ECAT Chemistry Chapter 6 Chemical Bonding
Try The MCQ's Test For ECAT Chemistry Chapter 6 Chemical Bonding
-
Total Questions30
-
Time Allowed30
Question # 1
During the formation of a chemical bond the potential energy of the system
Question # 2
Which of the following has highest bond order
Question # 3
The equation for the first ionization energy of hydrogen is
Question # 4
Which of the following statements is not correct regarding bonding molecular orbitals?
Question # 5
One of the following bonds is polar but compound is non-polar
Question # 6
Which of the following geometry is associated with the compound in which the central atom assumes sp3d hybridization?
Question # 7
The structure of ICI2is
Question # 8
The number of antibonding electron pairs in O2-molecular ion on the basis of MOT is
Question # 9
Nitrogen in NH3is sp3hybridized but the bond angle in NH3is 107° and not 109.5° due to
Question # 10
Which of the following molecules have multiple bonds
Question # 11
Mg becomes isoelectronic with neon when it
Question # 12
Atoms obey octet rule by sharing-electrons making covalent bonds according to
Question # 13
Antibonding MO is formed by
Question # 14
The geometry of 4 sp3hybrid orbitals on an atom is
Question # 15
The shape of the molecule SF2Cl2is
Question # 16
The number of bonds in nitrogen molecule is
Question # 17
Which for the following has no dipole moment
Question # 18
XeF4has shape of
Question # 19
An ionic compound M2S3is formed by the metal M,, the metal is
Question # 20
All covalent bonds formed between the two atoms are non-polar when
Question # 21
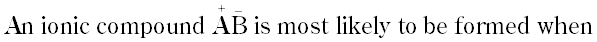
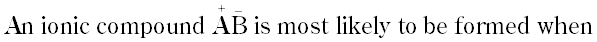
Question # 22


Question # 23
Generally the bond formed by metals with non-metals is
Question # 24
Generally ionization energy of atoms decreases by
Question # 25
Three sp2hybrid are co-planar at an angle of
Question # 26
The overlapping of two partially filled atomic orbital is in such a way that the probability of finding the electron pair is maximum along the axis joining the two nuclei, the bond is
Question # 27
The driving force for making a bond is
Question # 28
Water H2O is liquid while hydrogen sulphide H2S is a gas beause
Question # 29
The most suitable method of the separation of a mixture of ortho and para-nitrophenol mixed in the ratio of 1: 1 is
Question # 30
When of the following is isolelectronic with krypton
Top Scorers Of ECAT Chemistry Chapter 6 Chemical Bonding MCQ`s Test
-
T Tanzeel Ur Rehman 28 - May - 2024 04 Min 11 Sec 115/120 -
A Abdullah Zubair 20 - Dec - 2024 04 Min 30 Sec 115/120 -
M M Umer Arain 03 - Nov - 2024 00 Min 04 Sec 100/120 -
F Furqan Asif 10 - Mar - 2026 28 Min 39 Sec 100/120 -
M Manar Abbas 01 - Jun - 2024 04 Min 15 Sec 85/120 -
S Sania Iqbal 14 - May - 2024 06 Min 15 Sec 80/120 -
M Mirza Sibgha 27 - Jul - 2024 10 Min 11 Sec 80/120 -
G GOVT. SUFFA EDUCATION CENTER GIRLS MURIDKE 29 - May - 2024 13 Min 02 Sec 80/120 -
F Faisal Mehar 30 - May - 2024 16 Min 02 Sec 80/120 -
Z zakee 09u 30 - Nov - 2024 17 Min 43 Sec 80/120 -
N Noor Fatima 14 - Nov - 2024 06 Min 24 Sec 75/120 -
N Nayab Shahzad 08 - May - 2024 11 Min 21 Sec 70/120 -
M Muntasir Khokhar 21 - May - 2024 05 Min 39 Sec 65/120 -
N Naheed Yasir 26 - May - 2024 13 Min 03 Sec 65/120 -
S Shahid Baloxh 11 - May - 2024 14 Min 20 Sec 65/120
ECAT Chemistry Chapter 6 Important MCQ's
| Sr.# | Question | Answer |
|---|---|---|
| 1 | Which of the following has highest bond order | |
| 2 | Mg becomes isoelectronic with neon when it |
A. Loses two electrons
B. Gains two electrons
C. Loses 1 electron
D. Gains 1 electron
|
| 3 | Which of the hydrogen halides has the highest percentage of ionic character |
A. HCl
B. HBr
C. HF
D. HI
|
| 4 | The number of electron pairs shared in carbon tetrachloride molecule is |
A. 2
B. 3
C. 4
D. 1
|
| 5 | In which of the following theories the hybridizationis considered |
A. Vsepr
B. Lewis
C. Molecule orbital
D. Valence bond
|
| 6 | Ca, Mg, Be, Ba, belong to the same group, the order of their ionization energy values is |
A. Be > Mg> Ca > Ba
B. Ba > Ca > Mg > Be
C. Ca > Mg > Be > Ba
D. Ba > Mg > Ca > Be
|
| 7 | Electronegativity values of the elements F, CI and Br vary |
A. F > CI > Br
B. Br > CI > F
C. Cl > Br > F
D. CI > F > Br
|
| 8 | Which of the following has polar bond |
A. O2
B. N2
C. HCl
D. Cl2
|
| 9 | Which of the following molecules have sp3hybridized carbon |
A. CH4
B. C2H4
C. C2H2
D. CO2
|
| 10 | Elements have the tendency to attain 8 electrons in their valence shell. This is known as |
A. Octer rule
B. Hunds rule
C. Pauli exclusion principle
D. Auf ban principle
|








.gif)








Share your comments & questions here
No comments yet. Be the first to comment!