-
 Home
Home
-
 News
News
Latest Educational News Stories
Daily update of all national, international news, picture stories, college / university announcements and educational events.
-
 Colleges
Colleges
Pakistan's Largest Database of Colleges and Universities
Explore Largest Directory of Private and Govt. Colleges, Universities and find best institute for your future Education.
-
 Courses
Courses
-
 Admission
Admission
-
 Lectures
Lectures
-
 Online Test
Online Test
Short Question
- 9th Class Physics Short Questions
- 9th Class Chemistry Short Questions
- 9th Class Math Short Questions
- 9th Class Biology Short Questions
- 9th Class Computer Short Questions
- 9th Class English Short Questions
- 10th Class Physics Short Question
- 10th Class Chemistry Short Question
- 10th Class Math Short Question
- 10th Class Biology Short Question
- 10th Class Computer Short Question
- 10th Class English Short Question
-
 Past Papers
Past Papers
-
 Date Sheets
Date Sheets
-
 Results
Results
Exam Results 2024
Check online Results 2024 Matric Inter BA BSc B.Com MA MSc M.Com CSS PCS MCAT ECAT of all educational boards and universities in Pakistan
-
 Study Abroad
Study Abroad
Study Abroad Programs and Opportunities for Pakistani Students
Explore free study abroad search to find programs, consultants, events to study in USA, UK, Australia, China, Malaysia and many others.
-
 Jobs
Jobs
-
 Tutors
Tutors
-
 More
More
-
 Apps
Apps
MCQ's Test For ECAT Chemistry Chapter 18 Fundamental Principles of Organic Chemistry
Try The MCQ's Test For ECAT Chemistry Chapter 18 Fundamental Principles of Organic Chemistry
-
Total Questions30
-
Time Allowed30
Question # 1
A double bond consists of
Question # 2
Major component of natural gas is:
Question # 3
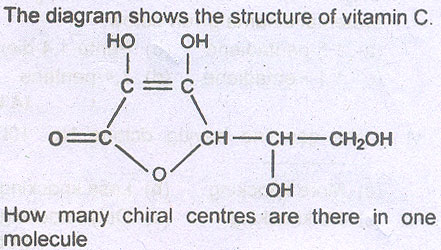

Question # 4
The major components of coal gas are
Question # 5
Alkanes normally have ______ hybridization
Question # 6
The process in which one s and two p orbitals mix up with each other is called
Question # 7
Which of the following has linear shape?
Question # 8
The state of hybridization of carbon atom in methane is
Question # 9
Which is the chain isomer of n-pentane
Question # 10
The general formula of saturated alicyclic hydrocarbons is:
Question # 11
Coal heated in the absence of air of about 500 - 1000°C is converted to
Question # 12
The formula of esters is
Question # 13
Source of organic compounds primarily is:
Question # 14
Which of the following is an electrophile
Question # 15
A nuclophile must
Question # 16
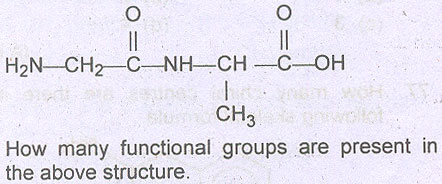

Question # 17
Angle 120° is observed in molecules :
Question # 18
Linear geometry is present in:
Question # 19
Normal by product of cracking is
Question # 20
Which is a mixture of low boiling hydrocarbon
Question # 21
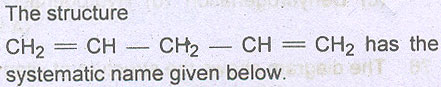
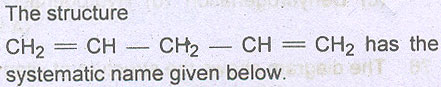
Question # 22
The self linking property of carbon is called as
Question # 23
Which of the following is complex?
Question # 24
Similarity in properties of different organic compounds give rise to the under standing of ______
Question # 25
Which of these always applies to a nucleophile
Question # 26
Cracking products are :
Question # 27
Wohler synthesized first of all the organic compound:
Question # 28
The quality of petroleum is determined by
Question # 29
Sp3 hybridization occurs when carbon is bound to
Question # 30
The open chain organic compounds are called
Top Scorers Of ECAT Chemistry Chapter 18 Fundamental Principles of Organic Chemistry MCQ`s Test
-
S Shazi Ansari 05 - Jun - 2024 01 Min 16 Sec 115/120 -
A Ahmad Chand 24 - Dec - 2024 01 Min 25 Sec 115/120 -
M M Umer Arain 25 - Nov - 2024 00 Min 07 Sec 110/120 -
A Abdul Rehman 21 - Jul - 2024 03 Min 05 Sec 110/120 -
U Usman Saif 29 - May - 2024 03 Min 46 Sec 95/120 -
F Furqan Asif 18 - Mar - 2026 38 Min 33 Sec 92/120 -
A Abdull Rehman 30 - May - 2024 05 Min 36 Sec 90/120 -
H HAFIZ MUHAMMAD HADEED ZAHID HAFIZ HADEED 08 - Dec - 2024 05 Min 52 Sec 90/120 -
G GOVT. SUFFA EDUCATION CENTER GIRLS MURIDKE 01 - Jun - 2024 08 Min 37 Sec 90/120 -
A Abdullah Akhtar 06 - Jun - 2024 14 Min 02 Sec 90/120 -
H Haider Ali 03 - Jun - 2024 14 Min 43 Sec 85/120 -
A Afzaal Falak 28 - Feb - 2026 06 Min 38 Sec 84/120 -
A Ayan Khan 06 - Aug - 2024 07 Min 32 Sec 80/120 -
F Fatima Anis 26 - Dec - 2024 06 Min 39 Sec 75/120 -
N Nasir Jamal 25 - Jan - 2026 06 Min 07 Sec 72/120
ECAT Chemistry Chapter 18 Important MCQ's
| Sr.# | Question | Answer |
|---|---|---|
| 1 | The general formula of saturated alicyclic hydrocarbons is: |
A. CnHn
B. CnH2n
C. CnH2n+2
D. CnH2n-1
|
| 2 | Select from the following the one which alcohol | |
| 3 | The atom other than C in a hetrocyclic compound is called as |
A. Hetroatom
B. Hetroion
C. Hetromolecule
D. Hetroelement
|
| 4 | Cracking done at low pressure and with a catalyst is called _______ cracking |
A. Thermal
B. Catalytic
C. Steam
D. None of them
|
| 5 | On passing ethane into concentrated suphuric acid the intermediate compound formed on hydrolysis with boiling water gives |
A. Methyl alcohol
B. Ethyl alcohol
C. Ethyl hydrogen sulphate
D. Methyl hydrogen sulphate
|
| 6 |

|
A. Addition Elimination
B. Addition Reduction
C. Elimination Reduction
D. Substitution Elimination
|
| 7 | Which of these always applies to a nucleophile |
A. It attacks a double bond
B. It has a lone pair of electrons
C. It is single atom
D. It is negatively charged
|
| 8 | The formula of esters is | |
| 9 | Type of isomerism in BrCH=CHBr is: |
A. Structural isomerism
B.
Conformational isomerism
C. Geometrical isomerism
D. Positional isomerism
|
| 10 | The quality of petroleum is determined by |
A. Decane number
B. Octane number
C. Nexane number
D. None of these
|








.gif)








Share your comments & questions here
Sabeen
13 Feb 2019How the result of the mcqs of that test I have solved here,I will get?