-
 Home
Home
-
 News
News
Latest Educational News Stories
Daily update of all national, international news, picture stories, college / university announcements and educational events.
-
 Colleges
Colleges
Pakistan's Largest Database of Colleges and Universities
Explore Largest Directory of Private and Govt. Colleges, Universities and find best institute for your future Education.
-
 Courses
Courses
-
 Admission
Admission
-
 Lectures
Lectures
-
 Online Test
Online Test
Short Question
- 9th Class Physics Short Questions
- 9th Class Chemistry Short Questions
- 9th Class Math Short Questions
- 9th Class Biology Short Questions
- 9th Class Computer Short Questions
- 9th Class English Short Questions
- 10th Class Physics Short Question
- 10th Class Chemistry Short Question
- 10th Class Math Short Question
- 10th Class Biology Short Question
- 10th Class Computer Short Question
- 10th Class English Short Question
-
 Past Papers
Past Papers
-
 Date Sheets
Date Sheets
-
 Results
Results
Exam Results 2024
Check online Results 2024 Matric Inter BA BSc B.Com MA MSc M.Com CSS PCS MCAT ECAT of all educational boards and universities in Pakistan
-
 Study Abroad
Study Abroad
Study Abroad Programs and Opportunities for Pakistani Students
Explore free study abroad search to find programs, consultants, events to study in USA, UK, Australia, China, Malaysia and many others.
-
 Jobs
Jobs
-
 Tutors
Tutors
-
 More
More
-
 Apps
Apps
MCQ's Test For ECAT Chemistry Chapter 11 Reaction Kinetics
Try The MCQ's Test For ECAT Chemistry Chapter 11 Reaction Kinetics
-
Total Questions30
-
Time Allowed30
Question # 1
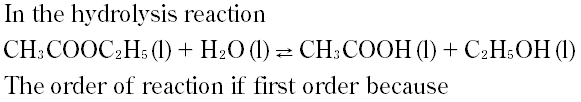
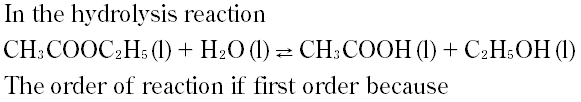
Question # 2
The unit of the rate constant is the same as that of rate of reaction in :
Question # 3
In the rate equation when the concentration of reactants are unity, then rate is equal to
Question # 4
The change in concentration of reactant or product per unit time is called :
Question # 5
The experimental relationship between a reaction rate and the concentration of reactants is called
Question # 6
In zero order reaction, the rate is independent of :
Question # 7
The unit of rate constant is the same as that of the rate of reaction in :
Question # 8
A white precipitate ofsilver chlorideimmediately formed on additionof :
Question # 9
______ are called biocatalysts
Question # 10
Which one of the following reaction rate is effected by the light
Question # 11
In the hydrolysis of CH3COOC2H5the acid produced is
Question # 12
Rate law of an equation is obtained :
Question # 13
With increases in temperature of 10 K of the reacting gases the rate of reaction is doubled because
Question # 14
which one of the following is a heteroheneous catalysis
Question # 15
Hydrolysis of ethyl-acetate (ester) has order of reaction :
Question # 16
The unit of rate of reaction is
Question # 17
A pseudo uni-molecular reaction has order of reaction :
Question # 18
The rate of reaction b/w two specific time intervals is called :
Question # 19
The factor which effect the rate of reaction
Question # 20
A zero order reaction is one in which :
Question # 21
All reactions occur in :
Question # 22
The value of activation energy Ea of a reaction can be determined from the value of slope of the straight line obtained by plotting a graph between 1/T and log k. the value of Ea is equal to
Question # 23
The rate of reaction :
Question # 24
Group l-A elements react with water fastly than the reaction of group ll-A elements because
Question # 25
In an experiment the concentration of a reactant 'A' is doubled the rate increases four times. If concentration in tripled, then rate increases nint times. Thus the rate is proportional to ______ of concentration of 'A'
Question # 26
To determine the rate of reaction chemically a graphical method is applied. A graph is plotted between the amount or reactant decomposed or product formed against the time. The rate dx/dtat any time is equal to
Question # 27
The actual number of atoms or molecules taking part in rate determining step is
Question # 28
The rate constant k of a reaction activation energy Ea and temperature are related by Arrhenius in the form of an equation which is
Question # 29
When we perform the same reaction by taking two different initial concentrations of a reactant for a second order reaction then
Question # 30
The rate of reaction :
Top Scorers Of ECAT Chemistry Chapter 11 Reaction Kinetics MCQ`s Test
-
A Abubakar basra 02 - Mar - 2024 05 Min 25 Sec 120/120 -
T Tanzeel Ur Rehman 29 - May - 2024 06 Min 39 Sec 115/120 -
M M Umer Arain 10 - Nov - 2024 00 Min 12 Sec 105/120 -
U Unaiza Usman 23 - Feb - 2024 04 Min 37 Sec 100/120 -
H Huzaifa Asim 28 - May - 2024 01 Min 53 Sec 95/120 -
G GOVT. SUFFA EDUCATION CENTER GIRLS MURIDKE 30 - May - 2024 05 Min 44 Sec 90/120 -
H Haseeb Ahmed 25 - Feb - 2024 10 Min 22 Sec 85/120 -
C Computer mail 31 - Mar - 2026 07 Min 26 Sec 84/120 -
B Bilawal Amjad 05 - Mar - 2024 23 Min 27 Sec 80/120 -
M Maryam Fatima 19 - Mar - 2024 05 Min 45 Sec 75/120 -
M Mustaqeem 28 - Feb - 2024 11 Min 57 Sec 75/120 -
T Tamshi Sohail 22 - Apr - 2024 20 Min 02 Sec 75/120 -
S Shazi Ansari 06 - Jun - 2024 01 Min 59 Sec 70/120 -
M Muntasir Khokhar 21 - May - 2024 06 Min 02 Sec 70/120 -
U uzma majeed 11 - Mar - 2024 06 Min 47 Sec 65/120
ECAT Chemistry Chapter 11 Important MCQ's
| Sr.# | Question | Answer |
|---|---|---|
| 1 | Rate of chemical reaction depends upon : |
A. The number of total collisions per second.
B. Number of molecules taking part in a chemical reaction.
C. Number of fruitful collisions per second
D. Number of fruitless collisions per second.
|
| 2 | Refrectrometric method is used when |
A. Reactions involving absorption of I.R. or U. V
B. Reactions involving change of refractive index
C. Reactions involving ions
D. Change of optical activity
|
| 3 | When initial concentration of reactants an order of reaction is given, then its half life period can be calculated by the equation | |
| 4 | Which of the following is not affected by light | |
| 5 | The rate constant k of a reaction activation energy Ea and temperature are related by Arrhenius in the form of an equation which is | |
| 6 | Which of the following may affect the rate constant (k) fro a reaction : |
A. Change in concentration.
B. Change in pressure.
C. Change in pH.
D. Change in temperature.
|
| 7 | In zero order reaction, the rate is independent of : |
A. Temperature of reaction
B. Concentration ofreactants
C. Concentration of products
D. None of these.
|
| 8 | Half life period of a reaction is inversely proportion to the initial concentration of the reactant, then order of reaction is |
A. Third order
B. Second order
C. Fist order
D. Zero order
|
| 9 |

|
A. Initial concentration of reaction
B. Initial concentration of products
C. Final concentration of products
D. Order of the reaction
|
| 10 | Half life period of a first order reaction is independent of: |
A. Presence of catalyst.
B. Conditions of temperature
C. Initial concentration of the compound
D. All of above
|








.gif)








Share your comments & questions here
No comments yet. Be the first to comment!