-
 Home
Home
-
 News
News
Latest Educational News Stories
Daily update of all national, international news, picture stories, college / university announcements and educational events.
-
 Colleges
Colleges
Pakistan's Largest Database of Colleges and Universities
Explore Largest Directory of Private and Govt. Colleges, Universities and find best institute for your future Education.
-
 Courses
Courses
-
 Admission
Admission
-
 Lectures
Lectures
-
 Online Test
Online Test
Short Question
- 9th Class Physics Short Questions
- 9th Class Chemistry Short Questions
- 9th Class Math Short Questions
- 9th Class Biology Short Questions
- 9th Class Computer Short Questions
- 9th Class English Short Questions
- 10th Class Physics Short Question
- 10th Class Chemistry Short Question
- 10th Class Math Short Question
- 10th Class Biology Short Question
- 10th Class Computer Short Question
- 10th Class English Short Question
-
 Past Papers
Past Papers
-
 Date Sheets
Date Sheets
-
 Results
Results
Exam Results 2024
Check online Results 2024 Matric Inter BA BSc B.Com MA MSc M.Com CSS PCS MCAT ECAT of all educational boards and universities in Pakistan
-
 Study Abroad
Study Abroad
Study Abroad Programs and Opportunities for Pakistani Students
Explore free study abroad search to find programs, consultants, events to study in USA, UK, Australia, China, Malaysia and many others.
-
 Jobs
Jobs
-
 Tutors
Tutors
-
 More
More
-
 Apps
Apps
MCQ's Test For ECAT Chemistry Chapter 10 Electrochemistry
Try The MCQ's Test For ECAT Chemistry Chapter 10 Electrochemistry
-
Total Questions30
-
Time Allowed30
Question # 1
In the electrolysis of fused bauxite (Al2O32H2O) with fused Cryolite (Na3AlF6) using carbon rods as anode. The product obtained at cathode is
Question # 2
The substance having highest conductivity at room temperature among the following is
Question # 3
Which statement is incorrect for balancing of redox reactions by ion-electron method
Question # 4
The best electrode used in salt bridge is KCl. Which other electrolyte an also be used for the purpose :
Question # 5
In electrolytic cells, the chemical changes may be :
Question # 6
In a solution of CuSO4how much time will be required to precipitate 2g copper by 0.5 ampere current?
Question # 7
The cathodic reaction in the electrolysis of dill H2SO4with Pt electrodes
Question # 8
In an electrolytic cell, the electrons flow from :
Question # 9
When electric current is used to carry out non-spontaneous redox, the process is called
Question # 10
In the reaction K2Cr2O7+ HCl + CrCl3+ Cl2+ H2O the element which is reduced is
Question # 11
Silver oxide battery has a voltage of
Question # 12
Reaction takes place at anode is
Question # 13
In the presence of copper electrodes the electrolysis of aqueous CuSO4produces at cathode
Question # 14
When quantity of electricity passed is one faraday then the mass deposited at the electrode is equal to
Question # 15
The reference electrode is made by using
Question # 16
The conductivity of strong electrolyte
Question # 17
Purification of an impure copper is made by electrolytic cell, in which impure copper is anode and pure copper is cathode, and the electrolyte used is
Question # 18
Prevention of corrosion of iron by Zn coating is called
Question # 19
Which statement is incorrect for NICAD battery
Question # 20
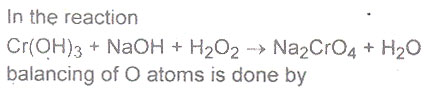
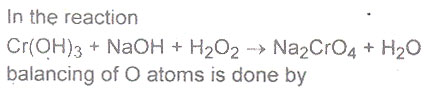
Question # 21
By using graphite electrode the electrolysis of aqueous solution of NaCI produces at anode
Question # 22
The oxidation number of free element is always taken to be
Question # 23
Out of Cu, Ag, Fe and Zn the metal which can displace all others from theri salt solution is
Question # 24
When aluminium electrode is coupled with copper electrode in a galvanic cell :
Question # 25
Which of the following (1 M) conducts more electricity?
Question # 26
If the standard electrode potential of CU2+/Cu electrode is 0.34 V, what is the electrode potential of 0.01 M concentration of Cu2+? (T=298)
Question # 27
A cell constant is generally found by measuring the conductivity of aqueous solution of
Question # 28
Calculate the amount of charge flowing in 2 minute in a wire of resistance 10Ωwhen a potential difference of 20 V is applied
Question # 29
The two half cells of a galvanic cell are connected by
Question # 30
When electrically is passed through molten Al2O3+ Na3AlF6and 13.5 gms of Al are deposited, the number of farady must be
Top Scorers Of ECAT Chemistry Chapter 10 Electrochemistry MCQ`s Test
-
A Abdul Ghaffar 06 - Jul - 2023 00 Min 03 Sec 120/120 -
U Unknown 08 - Jun - 2023 01 Min 18 Sec 120/120 -
A Abubakar basra 02 - Mar - 2024 05 Min 06 Sec 110/120 -
S Shazi Ansari 06 - Jun - 2024 02 Min 16 Sec 105/120 -
H Huzaifa Asim 28 - May - 2024 09 Min 10 Sec 100/120 -
U Unaiza Usman 22 - Feb - 2024 00 Min 35 Sec 95/120 -
O Omer Farooq 10 - Mar - 2023 09 Min 46 Sec 90/120 -
F Furqan Asif 21 - Mar - 2026 55 Min 45 Sec 80/120 -
A Amjid Ali 13 - Feb - 2024 10 Min 02 Sec 75/120 -
A Asim Akram 25 - Jan - 2024 04 Min 34 Sec 70/120 -
N Nasreen Sardar 08 - Jun - 2023 04 Min 44 Sec 70/120 -
A Arian Ahmed 06 - Mar - 2024 05 Min 06 Sec 65/120 -
A Abdullah Ashraf 01 - May - 2023 11 Min 07 Sec 65/120 -
T Tamshi Sohail 21 - Apr - 2024 13 Min 00 Sec 65/120 -
S Shahzaib Mujahid 23 - Jun - 2023 14 Min 50 Sec 65/120
ECAT Chemistry Chapter 10 Important MCQ's
| Sr.# | Question | Answer |
|---|---|---|
| 1 | In electronic watches or electronic calculators the tiny batteries used are |
A. Alkaline battery
B. NICAD battery
C. Fuel cell
D. Silver oxide battery
|
| 2 | The cathodic reaction in the electrolysis of dill H2SO4with Pt electrodes |
A. Reduction
B. Oxidation
C. Both oxidation and reduction
D. neither oxidation or reduction
|
| 3 | The galvanic or voltaic cells which are rechargeable called as |
A. Primary cells
B. Secondary cells
C. Dry cells
D. Infinite cells
|
| 4 | During electrolysis, the reaction that takes place at cathode is : |
A. Reduction
B. Both (a) and (c)
C. Oxidation
D. No reaction occurs
|
| 5 | An electric current is passed through silver nitrate solution using silver electrodes.10.79 g of silver was found to be deposited on the cathode if the same amount of electricity is passed through copper sulphate solution using copper electrodes, the weight of copper deposited on the cathode is |
A. 6.4 g
B. 2.3 g
C. 128.8 g
D. 3.2 g
|
| 6 | Electrolytes in the form of solution or in the fused state have the ability to conduct : |
A. Light.
B. Electricity.
C. Ions.
D. Electrons.
|
| 7 | Sodium metal is obtained by the electrolysis of fused NaCl in cell is called |
A. Nelson's cell
B. Down's cell
C. Daniell cell
D. Voltaic cell
|
| 8 | Which one of the following reaction takes place spontaneously | |
| 9 | Most metals are conductors of electricity because of the : |
A. Light weight.
B. Immobility of the electrons.
C. Lustrous surfaces
D. Relatively free movement of their electrons
|
| 10 | What will be the weight of deposited silver on passing 965 coulombs of electricity in solution of AgNO3? |
A. 1.08 g
B. 2.16 g
C. 0.54 g
D. 0.27 g
|








.gif)








Share your comments & questions here
No comments yet. Be the first to comment!