-
 Home
Home
-
 News
News
Latest Educational News Stories
Daily update of all national, international news, picture stories, college / university announcements and educational events.
-
 Colleges
Colleges
Pakistan's Largest Database of Colleges and Universities
Explore Largest Directory of Private and Govt. Colleges, Universities and find best institute for your future Education.
-
 Courses
Courses
-
 Admission
Admission
-
 Lectures
Lectures
-
 Online Test
Online Test
Short Question
- 9th Class Physics Short Questions
- 9th Class Chemistry Short Questions
- 9th Class Math Short Questions
- 9th Class Biology Short Questions
- 9th Class Computer Short Questions
- 9th Class English Short Questions
- 10th Class Physics Short Question
- 10th Class Chemistry Short Question
- 10th Class Math Short Question
- 10th Class Biology Short Question
- 10th Class Computer Short Question
- 10th Class English Short Question
-
 Past Papers
Past Papers
-
 Date Sheets
Date Sheets
-
 Results
Results
Exam Results 2024
Check online Results 2024 Matric Inter BA BSc B.Com MA MSc M.Com CSS PCS MCAT ECAT of all educational boards and universities in Pakistan
-
 Study Abroad
Study Abroad
Study Abroad Programs and Opportunities for Pakistani Students
Explore free study abroad search to find programs, consultants, events to study in USA, UK, Australia, China, Malaysia and many others.
-
 Jobs
Jobs
-
 Tutors
Tutors
-
 More
More
-
 Apps
Apps
MCQ's Test For ECAT Chemistry Chapter 1 Basic Concepts
Try The MCQ's Test For ECAT Chemistry Chapter 1 Basic Concepts
-
Total Questions30
-
Time Allowed30
Question # 1
The largest number of molecules are present in
Question # 2
The pressure of vapours when sent to the ionization chamber in mass spectrometer is
Question # 3
NH3, HCL, H2O, HL are:
Question # 4
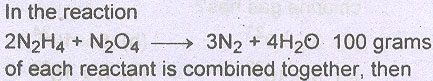
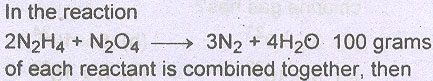
Question # 5
The branch of science dealing with structure, composition and changes in matter and laws and principles which govern these changes is called as
Question # 6
Which statement about molecule is incorrect ?
Question # 7
Which one of the following statements is not correct
Question # 8
A ring contains 3 gram diamond. The number of C-atoms which a ring contains is
Question # 9
Which of the sub-atomic particles is not charged
Question # 10


Question # 11
The empirical formula of a liquid compound is known to be C2H4O. What other information is needed to work out its molecular formula?
Question # 12
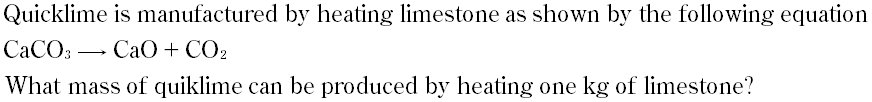
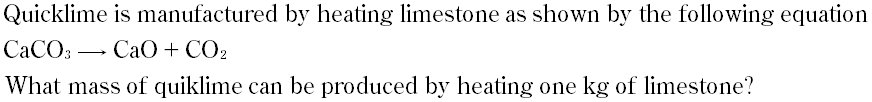
Question # 13
Which of the following statements is not true?
Question # 14
Atoms can be evident by use of electron microscope, field ionization microscope and:
Question # 15
A limiting reactant is the one which
Question # 16


Question # 17
Hemoglobin contains nearly:
Question # 18
Relative atomic mass of an element is the mass of the element relative to
Question # 19
Al3+is a symbol for aluminium
Question # 20
The number of isotopes of gold is
Question # 21
Mass spectrometer measures the
Question # 22
A molecule of haemoglobin is made up if nearly
Question # 23
A beaker contains 9 grams of water. The number of H-atoms is
Question # 24
How many moles of hydrogen atoms does 3.2 g of methane, CH4, contain?
Question # 25
The number of moles of CO2which contain 8.0 g of oxygen
Question # 26
0.5 mole of CH4and 0.5 mole of SO2gases have equal
Question # 27
Isotopes differ in
Question # 28
CO+ is an example of
Question # 29
Isotopes differ in the
Question # 30
Isotopes of an element differ in
Top Scorers Of ECAT Chemistry Chapter 1 Basic Concepts MCQ`s Test
-
F Fatima Tabassum 09 - Jan - 2025 04 Min 47 Sec 100/120 -
Z zakee 09u 28 - Nov - 2024 18 Min 56 Sec 95/120 -
H habib rehman 17 - Apr - 2026 24 Min 01 Sec 92/120 -
A Aroosha Tariq 13 - Nov - 2024 06 Min 02 Sec 90/120 -
Z Zainab Tanvir 07 - Dec - 2025 09 Min 10 Sec 80/120 -
I ISI?Shadow 16 - Nov - 2024 17 Min 47 Sec 80/120 -
N notghouri playz 13 - Dec - 2024 27 Min 04 Sec 80/120 -
T Tayyaba Attique 16 - Nov - 2024 07 Min 22 Sec 75/120 -
U umerasif 14 - Nov - 2024 105 Min 42 Sec 75/120 -
M Muhammad Zain 25 - Apr - 2026 09 Min 22 Sec 72/120 -
A agha hamza 22 - Nov - 2025 22 Min 17 Sec 72/120 -
A abdullah masood 04 - Dec - 2024 10 Min 54 Sec 70/120 -
M Muhammad Awais 01 - Jan - 2025 05 Min 43 Sec 65/120 -
A Aqsa afridi 02 - Dec - 2024 24 Min 01 Sec 65/120 -
A afraz afraz 17 - Jan - 2025 00 Min 39 Sec 60/120
ECAT Chemistry Chapter 1 Important MCQ's
| Sr.# | Question | Answer |
|---|---|---|
| 1 |

|
A. 8 g
B. 16 g
C. 32 g
D. 24 g
|
| 2 | Atoms can be evident by use of electron microscope, field ionization microscope and: |
A. x-rays
B. Video camera
C. Telescope
D. Compound microscope
|
| 3 |

|
A. 32 g
B. 3.2 g
C. 5.6 g
D. 9.6 g
|
| 4 | Who one mole of each of the following is completely burned in oxygen, which gives the largest mass of carbon dioxide? |
A. Carbon monoxide
B. Diamond
C. Ethane
D. Methane
|
| 5 | Two different hydrocarbon each contain the same percentage by mass of hydrogen. It follows that they have the same |
A. Empirical formula
B. Number of atoms in a molecules
C. Number of isomers
D. Relative molecular mass
|
| 6 | Each molecule of haemoglobin is 68000 times heavier than one atom of |
A. C
B. H
C. N
D. O
|
| 7 |
The diameter of atoms is of the order: |
A. 2* 10-5m
B.
2* 10-10 m
C. 2* 10-2m
D. 2* 10-3m
|
| 8 | When 0.1 g of magnesium is treated with an excess of hydrochloric acid, what volume of gas at room temperature and pressure will be produced |
A. 10 cm3
B. 25 cm3
C. 48 cm3
D. 100 cm3
|
| 9 | Objects of the size of an atom can be observed in |
A. An electron microscope
B. An x-ray spectrum
C. Atomic absorption spectrum
D. A visible spectrum
|
| 10 | Benzene is stable to: |
A. Oxidation
B. Nitration
C. KMnO4
D. SULPHONATION
|








.gif)








Share your comments & questions here
ishaq
05 Mar 2019where is the syllabus or preparation material
ishaq
05 Mar 2019...
Mehwish
30 Jun 2018...